Derivation of the Relationship Between Half-Life Constants This means that the fossil is 11,460 years old. 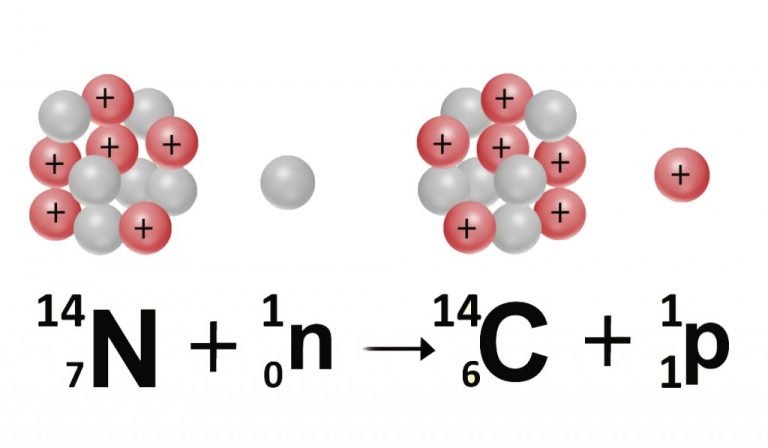
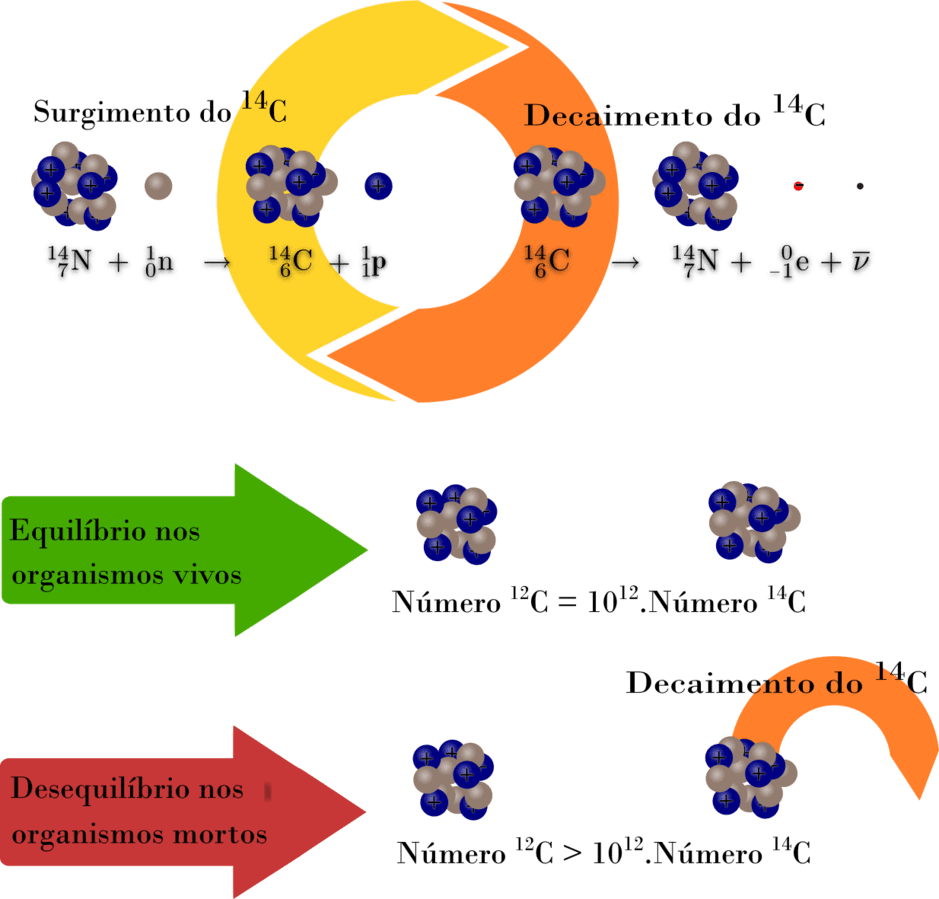
If an archaeologist found a fossil sample that contained 25% carbon-14 in comparison to a living sample, the time of the fossil sample's death could be determined by rearranging equation 1, since N t, N 0, and t 1/2 are known. N t is the remaining quantity after time, t The carbon-14 undergoes radioactive decay once the plant or animal dies, and measuring the amount of carbon-14 in a sample conveys information about when the plant or animal died.īelow are shown three equivalent formulas describing exponential decay: It is incorporated into plants through photosynthesis, and then into animals when they consume plants. The process of carbon-14 dating was developed by William Libby, and is based on the fact that carbon-14 is constantly being made in the atmosphere. The half-life of carbon-14 is approximately 5,730 years, and it can be reliably used to measure dates up to around 50,000 years ago. One of the most well-known applications of half-life is carbon-14 dating. The term is most commonly used in relation to atoms undergoing radioactive decay, but can be used to describe other types of decay, whether exponential or not. Or even look at GCSE level questions on this topic.Half-life is defined as the amount of time it takes a given quantity to decrease to half of its initial value. Or questions relating to practical investigations You may also want to try Rdioactivity Questions The counts are corrected for background radiation.Ĭalculate the age, in years, of the archaeological samples of bone. The proportion of 14C is constant in living organisms.Ī 1 g sample of bone from an archaeological site has an average rate of decay of 5.2 Bq due to 14C.Ī 1 g sample of bone from a modern skeleton has a rate of decay of 6.5 Bq. 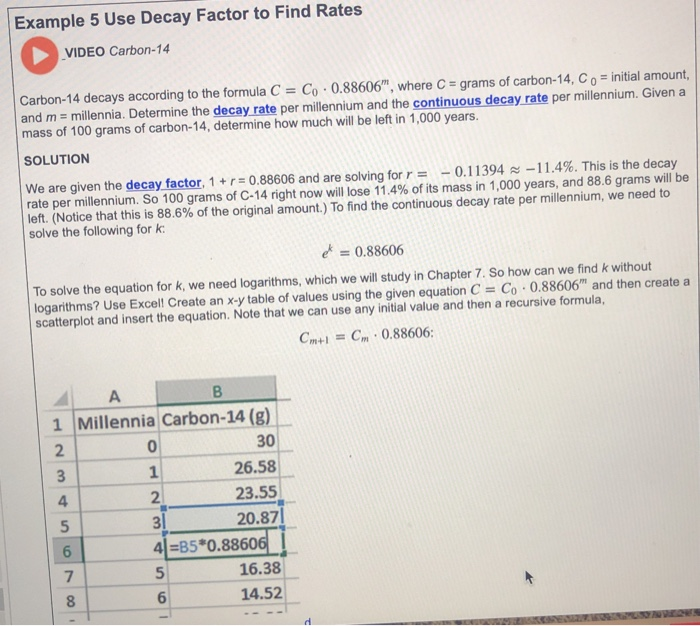
The age of a piece of bone recovered from an archaeological site may be estimated by 14C dating.Īll living organisms absorb 14C but there is no further intake after death. (e) Give two reasons why it is difficult to obtain a reliable age of the ancient boat from the carbon dating described. The rate of decay due to the atoms in this sample is 0.65 Bq.Ĭalculate the age of the ancient boat in years. (d) A sample of 3.00 × 10 23 atoms of carbon is removed from a piece of wood taken from the ancient boat. (c) Show that the rate of decay of the atoms in the living wood sample is 1.15 Bq. (b) Calculate the half-life of the wood in years, giving your answer to an appropriate number of significant figures. In living wood one in 10 12 of the carbon atoms is of the radioactive isotope, which has a decay constant of 3.84 × 10 –12 s –1. The age of an ancient boat may be determined by comparing the radioactive decay of from living wood with that of wood taken from the ancient boat.Ī sample of 3.00 × l0 23 atoms of carbon is removed for investigation from a block of living wood. (b) Suggest why the method of carbon dating is likely to be unreliable if a sample is: (ii) A piece of wood taken from an axe handle found on an archaeological site has 0.375 times as many carbon-14 atoms as an equal mass of living wood.Ĭalculate the age of the axe handle in years. (i) The half-life of carbon-14 is 5740 years.Ĭalculate the radioactive decay constant in yr −1 of carbon-14. After a tree dies, the proportion of carbon-14 in it decreases due to radioactive decay. The carbon content of living trees includes a small proportion of carbon-14, which is a radioactive isotope.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
